|
GNC502S - GENERAL CHEMISTRY 1B -2ND OPP - JAN 2023 |
 |
1 Page 1 |
▲back to top |

 |
2 Page 2 |
▲back to top |

 |
3 Page 3 |
▲back to top |

 |
4 Page 4 |
▲back to top |

 |
5 Page 5 |
▲back to top |

 |
6 Page 6 |
▲back to top |

 |
7 Page 7 |
▲back to top |

 |
8 Page 8 |
▲back to top |

 |
9 Page 9 |
▲back to top |

 |
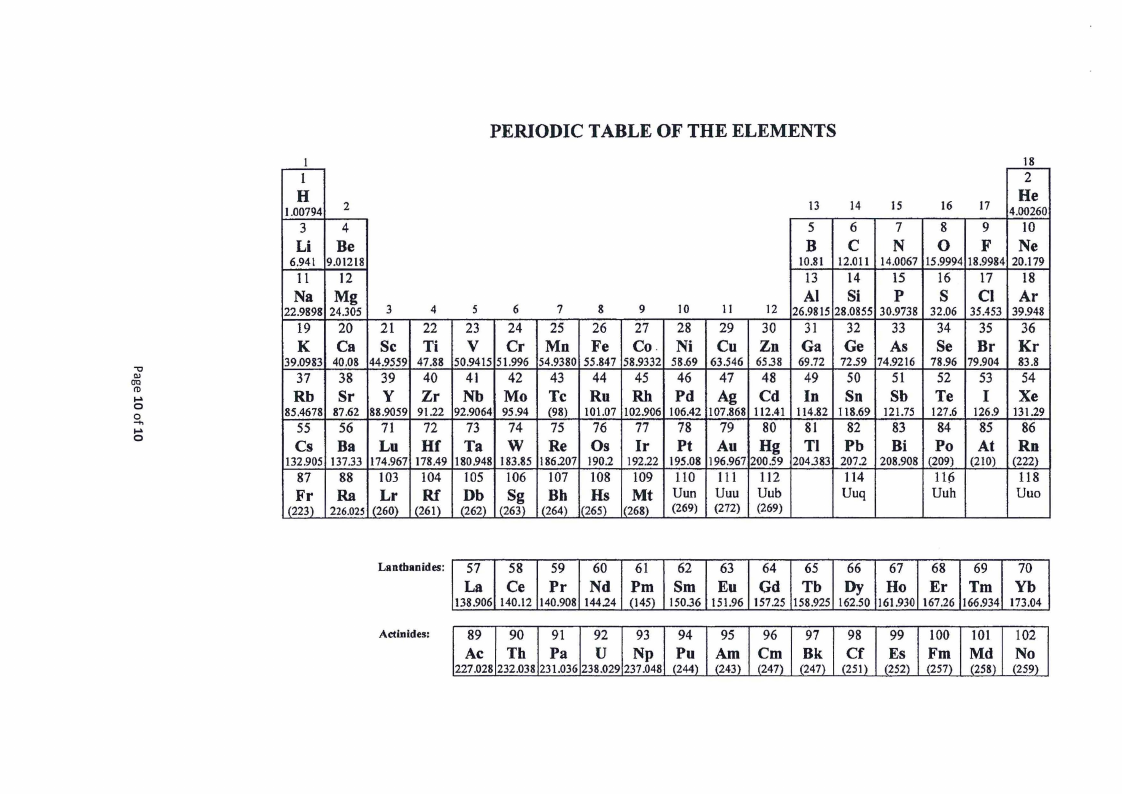
10 Page 10 |
▲back to top |