 |
ICA511S - INTRODUCTION TO CHEMISTRY - 1ST OPP - JUNE 2025 |
 |
1 Page 1 |
▲back to top |

nAml BIA un,vERSITY
OF SCIEnCEAno TECHn OLOGY
FACULTYOF HEALTH,NATURALRESOURCESAND APPLIEDSCIENCES
SCHOOLOF AGRICULTUREAND NATURALRESOURCESSCIENCES
DEPARTMENT OF AGRICULTURALSCIENCEAND AGRIBUSINESS
QUALIFICATIONS:BACHELOROF SCIENCEIN AGRICULTURE
BACHELOROF SCIENCEIN HORTICULTURE
QUALIFICATIONSCODE: 07BAGA LEVEL: NQF LEVEL7
07BHOR
COURSECODE: ICA511S
COURSENAME: INTRODUCTION TO CHEMISTRY
DATE: JUNE 2025
SESSION:JUNE
DURATION: 3 HOURS
MARKS: 100
EXAMINER:
MODERATOR:
FIRSTOPPORTUNITY EXAMINATION QUESTION PAPER
MS. PAULINA NDINELAGO NAUPU
MRS. LUCIA TUYENI-KELAO KAFIDI
INSTRUCTIONS
1. Answer all the questions.
2. Write neatly and clearly.
3. Mark all answers clearly with their respective question numbers.
4. All written work MUST be done in blue or black ink.
5. No books, notes and other additional aids are allowed.
PERMISSIBLEMATERIALS
1. Calculator
2. Examination paper
3. Examination script
THIS MARKING SCHEMECONSISTSOF 3 PAGES
(ExcludingThis Front Page)
0
 |
2 Page 2 |
▲back to top |

QUESTION 1
Define the following terms.
1.1 Atoms
1.2 Conversion factor
1.2 Element
1.3 Matter
1.4 Molecules
1.5 Compound
1.6 Homogeneous mixture
{2}
{2}
{2}
{2}
{2}
{2}
[12]
QUESTION 2
2.1 State the four Dalton's theory and give an example of each theory. {8}
2.2 Fully describe the concept of an ionic bond.
{5}
2.3 How are molarity and molality related in terms of concentration measurements?
{3}
2.4 Explain Avogadro's number in detail, including its definition, significance in
chemistry, and how it is used in calculations. Provide its numerical value
{5}
[21]
QUESTION 3
3.1
Imagine you want to measure out 0.214 ml of benzene but lack the equipment
to accurately measure such a small volume. You are, however, equipped with an
analytical balance capable of measuring up to ±0.000lg. Looking at a reference
table, you learns the density of benzene (p=0.8765g/ml}. How many grams of
benzene should you use? Use the calculation rules and give your final answer to
the correct significant figure
{5}
3.2 A cyclist is riding at a speed of 5 km/hr. To analyze their motion in a physics
experiment, you need to express this speed in meters per second (m/s). Using
the appropriate conversion factors, determine the cyclist's speed in m/s and
round your final answer to one significant figure.
{6}
QUESTION 4
4.1 A chemist measures 78.5 g of aluminum
aluminum are present.
(Al). Calculate how
[11]
many moles of
{4}
4.2 How many grams of calcium carbonate (CaCO3}are present in 0.75 moles?
{5}
1
 |
3 Page 3 |
▲back to top |

4.3 A farmer adds 156 g of ammonium sulfate ((NH4}zSO4)to the soil. Determine the
number of ammonium sulfate molecules in the sample.
{8}
4.4 A student dissolves 68.4 g of an unknown substance in water and determines that
the solution contains 1.2 moles of the substance. Calculate the molar mass and
suggest a possible identity based on common molar masses.
{5}
[22)
QUESTION 5
5.1 Calculate the molarity of a solution made by dissolving 23.4 g of sodium sulfate
(Na2SO4)in enough water to form 125 ml of solution.
{9}
5.2 If a compound has an empirical formula of CH2 and a molar mass of 84 g/mol,
what is its molecular formula
{5}
[14)
QUESTION 6
6.1 Calculate the percentage composition of carbon in these substances.
{8}
6.2 2 moles of propane react with 8 moles of oxygen gas in a combustion reaction in
the following equation: lC3Hs +
3CO2+ 4H2O. What is the limiting reactant
{5}
6.3 30g of C3Hsburns in air to produce 70g of CO2using the following reaction C3Hs+
502 3CO2+4H2O. Calculate the theoretical and percentage yield.
{4}
6.4 Calculate the percent yield
{3}
[20)
Total Marks:
100
2
 |
4 Page 4 |
▲back to top |
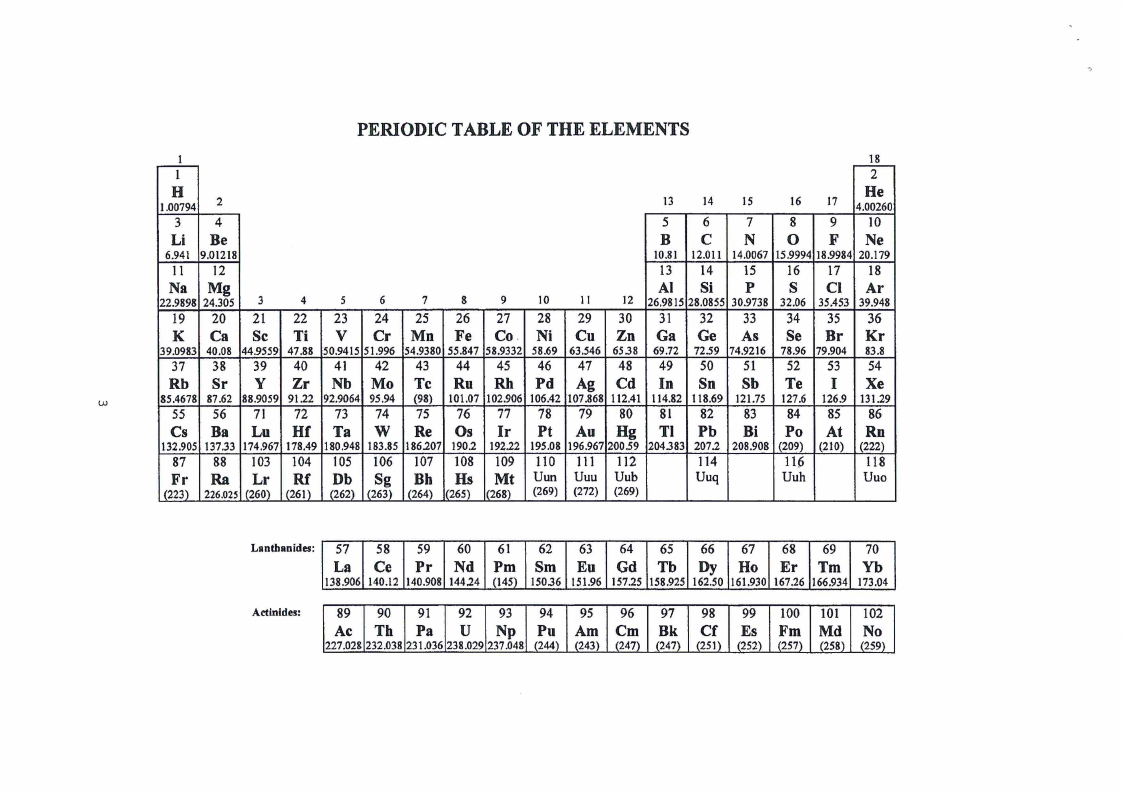
PERIODICTABLEOF THE ELEMENTS
-
18
l
2
H
).00794 2
13
14
15
He
16
17 4.00260
34
5
6
7
8 9 10
Li Be
B C N 0 F Ne
6.941 9.01218
JO.SI 12.011 14.0067 15.9994 18.9984 20.179
I I 12
13 14 15 16 17 18
Na Mg
Al Si p s CI Ar
22.9898 24.305 3
4
5
6
7
8
9
10
11
12 26.9815 28.0855 30.9738 32.06 35.453 39.948
19 20 21 22 23 24 25 26 27 28 29 30 31 32 33 34 35 36
K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn Ga Ge As Se Br Kr
39.0983 40.08 44.9559 47.88 50.9415 51.996 54.9380 55.847 58.9332 58.69 63.546 6S38 69.72 72.59 74.9216 78.96 79.904 83.8
37 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54
Rb Sr y Zr Nb Mo Tc Ru Rh Pd Ag Cd In Sn Sb Te I Xe
w
85.4678 87.62 88.9059 91.22 92.9064 95.94 {98) 101.07 102.906 106.42 107.868 112.41 114.82 118.69 121.75 127.6 126.9 131.29
55 56 71 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86
Cs Ba Lu Hf Ta w Re Os Ir Pt Au Hg Tl Pb Bi Po At Rn
132.90S 137.33 174.967 178.49 180.948 183.85 186.207 190.2 192.22 195.08 196.967 200.59 204383 207.2 208.908 {209) (210) (222)
87 88 103 104 105 106 107 108 109 110 111 IJ2
114
116
118
Fr Ra Lr Rf Db Sg Bh Hs Mt Uun Uuu Uub
Uuq
(223) 226.025 (260) (261) (262) (263) (264) (265) (268) (269) (272) (269)
Uuh
Uuo
Lanthanides: 57 58 59 60 61 62 63 64 65 66 67 68 69 70
La Ce Pr Nd Pm Sm Eu Gd Tb Dy Ho Er Tm Yb
138.906 140.12 140.908 144.24 (145) 15036 151.96 157.25 158.925 162.50 161.930 167.26 166.934 173.04
Actinides:
89 90 91 92 93 94 95 96 97 98 99 100 101 102
Ac Th Pa u Np Pu Am Cm Bk Cf Es Fm Md No
227.028 232.038 231.036 238.029 237.048 (244) (243) (247) (247) (251) (252) (257) (258) (259)





